
The results of two phase III clinical trials showed that at the 14th week, whether in patients with non radiologic axial spondyloarthritis (nr axSpA) or in patients with active Ankylosing spondylitis (AS) with poor biological treatment effect, compared with placebo, more patients in the Upatinib group reached the ASAS 40 response. The patient's symptoms have achieved statistically significant relief, including back pain and inflammation, as well as improvements in physical function and disease activity. Axial spinal arthritis is a chronic inflammatory disease that affects the spine, causing back pain, limited mobility, and structural damage.
In patients with active Ankylosing spondylitis (AS)
45% of patients in the Upatinib group met the ASAS40 standard, significantly better than the placebo group (18%).
In addition, Upatinib also achieved multiple secondary endpoints of the experiment, as shown in the following figure.
The safety data is consistent with previous studies and no new risks have been identified.
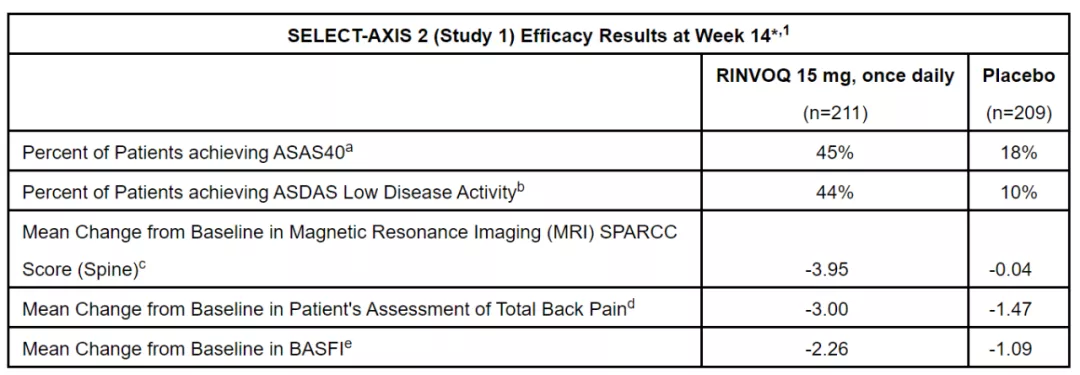
Image source: Reference [1]
In patients with active non radiographic axial spinal arthritis
45% of patients in the Upatinib group met the ASAS40 standard, significantly higher than the placebo group (23%).
In addition, Upatinib also achieved multiple secondary endpoints of the experiment, as shown in the following figure.
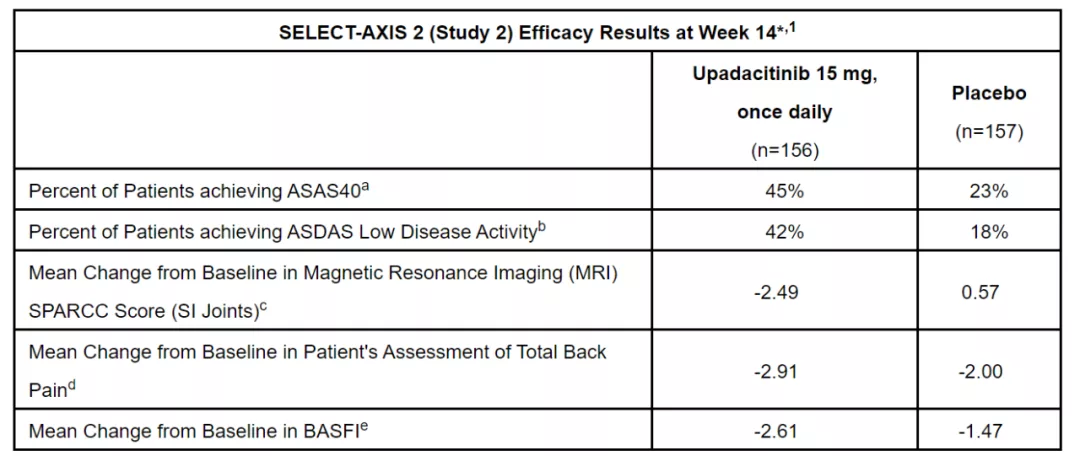
Image source: Reference [2]
"Ankylosing spondylitis is a debilitating disease, which can cause severe pain, stiffness, limited activity and lasting structural damage, and seriously affect the daily life of patients." Dr. Michael Severino, vice chairman and president of AbbVie, said, Similarly, patients with active non radiological axial osteoarthritis may experience chronic inflammatory back pain and limited physical function. These patients require treatment to help improve their physical function and quality of life. We are encouraged by the potential of Upatinib for meaningful disease control and relief of these symptoms
Upatinib, discovered and developed by AbbVie scientists, is a selective and reversible JAK inhibitor that is taken orally once a day. In August 2019, it was approved by the US FDA for the treatment of adult patients with moderate to severe active rheumatoid arthritis who have insufficient or intolerant response to methotrexate. The JAK protein family mediates the signaling of various inflammatory factors. Upatinib has achieved positive results in the phase III clinical trials for the treatment of Rheumatoid arthritis, atopic dermatitis, Crohn's disease, psoriatic arthritis and other inflammatory diseases.
参考资料:[1] AbbVie's RINVOQ® (upadacitinib) Met Primary and All Ranked Secondary Endpoints in Phase 3 Study in Ankylosing Spondylitis. Retrieved October 7, 2021, from https://www.prnewswire.com/news-releases/abbvies-rinvoq-upadacitinib-met-primary-and-all-ranked-secondary-endpoints-in-phase-3-study-in-ankylosing-spondylitis-301395108.html[2] AbbVie's upadacitinib (RINVOQ®) Met Primary and Most Ranked Secondary Endpoints in Phase 3 Study for Non-Radiographic Axial Spondyloarthritis. Retrieved October 7, 2021, from https://www.prnewswire.com/news-releases/abbvies-upadacitinib-rinvoq-met-primary-and-most-ranked-secondary-endpoints-in-phase-3-study-for-non-radiographic-axial-spondyloarthritis-301395109.html
Disclaimer: WuXi AppTec content team focuses on introducing global biomedical health research progress. This article is only for the purpose of information exchange. The views in this article do not represent WuXi AppTec's position, nor do they represent WuXi AppTec's support or opposition to the views in this article. This article is not a recommended treatment plan. If you need guidance on treatment plans, please go to a reputable hospital for medical treatment.
Copyright Notice: Lianchuang Biomedical publishes original and reprinted articles. The purpose of reprinting articles is to recommend valuable information and share it with more friends. We respect intellectual property rights, are committed to protecting intellectual property rights, and do our best to indicate the author and source. If there are copyright issues with the articles, images, and other content we reprint, please contact us at 0551-68596228. We will do our best to contact you as soon as possible and handle them promptly. Sincerely thank you for your understanding and assistance!
 Home
Home
 Product
Product
 News
News
 Contact
Contact